SEOUL, Aug. 7 (Korea Bizwire) – South Korean artificial intelligence (AI) healthcare companies are making significant progress in the global market, with recent developments highlighting their competitiveness and potential for expansion.
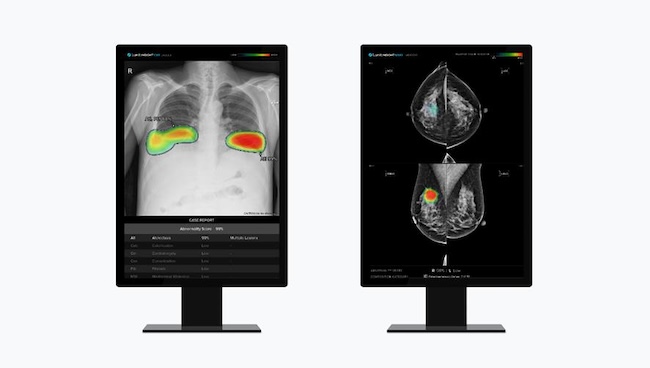
Lunit, a leader in AI-powered cancer diagnostics, has announced that its chest X-ray analysis solution, Lunit Insight CXR, has received approval from the World Health Organization (WHO) for use in tuberculosis screening programs.
This marks the first time an AI-based chest X-ray diagnostic aid has been included in the WHO’s list of recommended products for systematic screening of tuberculosis.
Suh Beomseok, CEO of Lunit, stated, “This approval from the WHO validates Lunit’s competitiveness in the global healthcare market. We plan to actively expand our market presence, particularly in developing countries, based on this achievement.”
In another development, DeepBio, an AI-powered cancer diagnosis company, has reported that its AI prostate cancer analysis solution, DeepDx Prostate, has been validated for diagnostic accuracy in collaboration with Stanford University School of Medicine.
The results, published in the British Journal of Urology, showed that the solution achieved a sensitivity of 0.997 and a specificity of 0.88 when diagnosing specimens obtained through radical prostatectomy.
Kim Sun Woo, CEO of DeepBio, emphasized the potential impact of this technology: “Pathological examination of prostate cancer surgical specimens requires significant time and effort. Utilizing AI analysis algorithms for prostate cancer in clinical settings could lead to notable improvements in both efficiency and performance of pathological examinations.”
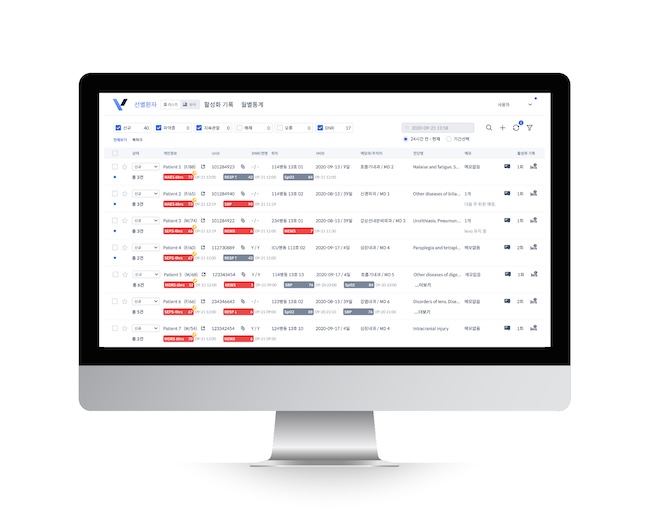
The trend of South Korean AI healthcare companies gaining international recognition is further exemplified by AITRICS, which recently obtained 510(k) pre-market approval from the U.S. Food and Drug Administration (FDA) for its inpatient monitoring solution, VitalCare (AITRICS-VC).
The 510(k) is an FDA process that verifies the equivalence of a new medical device with previously approved devices before it can be marketed.
VitalCare is designed to help medical professionals predict the risk of emergency situations early by analyzing patients’ vital signs and blood test results.
This follows a similar FDA approval received by JLK in June for its AI-powered prostate cancer diagnosis solution, Medihub Prostate.
Kevin Lee (kevinlee@koreabizwire.com)